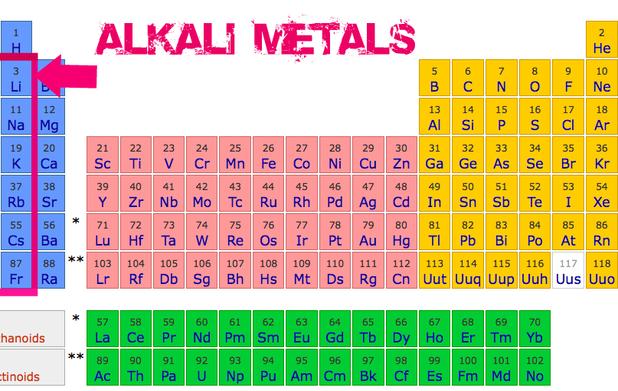
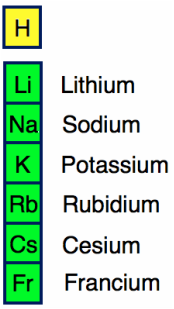
Alkali Metals
Alkali metals are group 1 of the periodic table excluding Hydrogen since it's nonmetal. They are the most active metals along with alkaline earth metals. They react strongly with "water to release a hydrogen gas"(The Elements), creating an explosion. Alkali metals have a valence electron of 1 and have a large radius(size). They also have a low ionization energy level and a low electronegativity level. The alkali metals are Lithium(Li), Sodium(Na), Potassium(K), Rubidium(Rd), Cesium(Cs), and Francium(Fr). The elements in group 1 also begin every period.
Information about the Alkali Metals
Lithium(Li) is element #3 on periodic table and is the least reactive of the alkali metals. [Its ionization energy level is 520 kJ/mol, electronegativity level is 1.0, and the atomic radius is 130 pm.*] Lithium is used in lithium batteries and lithium carbonate pills, which are used to control mood swings.
Sodium(Na) is element #11 and begins period 3. [Ionization energy:496 kJ/mol, electronegativity level: 0.9, and atomic radius: 160 pm.*] Sodium is used in lye, engine valves, hollow cathode lamps, and Himalayan sea salt.
Potassium(K) is element #19 and starts period 4. [IE:419 kJ/mol, electronegativity level: 0.8, and atomic radius: 200 pm.*] Potassium is used in cooking alum, salt substitute, and bananas!
Rubidium(Rb) is element #37 and starts period 5. [IE:403 kJ/mol, electronegativity level: 0.8, and atomic radius: 215 pm.*] It is used in rubidium time standard exciter, rubidium clock cells, and rubidium vapor cells.
Cesium(Cs) is element #55 and begins period 6. [IE: 376 kJ/mol, electronegativity level: 0.9, and atomic radius: 238 pm.*] It is used for time. There are 300 synchronized Cesium clocks around the world, to keep the universal time. It is also used in Himalayan sea salt and in the clocks that are around the world.
Francium(Fr) is element #87 and begins the last row, period 7. It is considered the most reactive of the alkali metals. Francium is radioactive and its name is derived from the country, France.
*All informational data is from Reference Tables for Physical Setting: CHEMISTRY-2011 edition NY
Lithium(Li) is element #3 on periodic table and is the least reactive of the alkali metals. [Its ionization energy level is 520 kJ/mol, electronegativity level is 1.0, and the atomic radius is 130 pm.*] Lithium is used in lithium batteries and lithium carbonate pills, which are used to control mood swings.
Sodium(Na) is element #11 and begins period 3. [Ionization energy:496 kJ/mol, electronegativity level: 0.9, and atomic radius: 160 pm.*] Sodium is used in lye, engine valves, hollow cathode lamps, and Himalayan sea salt.
Potassium(K) is element #19 and starts period 4. [IE:419 kJ/mol, electronegativity level: 0.8, and atomic radius: 200 pm.*] Potassium is used in cooking alum, salt substitute, and bananas!
Rubidium(Rb) is element #37 and starts period 5. [IE:403 kJ/mol, electronegativity level: 0.8, and atomic radius: 215 pm.*] It is used in rubidium time standard exciter, rubidium clock cells, and rubidium vapor cells.
Cesium(Cs) is element #55 and begins period 6. [IE: 376 kJ/mol, electronegativity level: 0.9, and atomic radius: 238 pm.*] It is used for time. There are 300 synchronized Cesium clocks around the world, to keep the universal time. It is also used in Himalayan sea salt and in the clocks that are around the world.
Francium(Fr) is element #87 and begins the last row, period 7. It is considered the most reactive of the alkali metals. Francium is radioactive and its name is derived from the country, France.
*All informational data is from Reference Tables for Physical Setting: CHEMISTRY-2011 edition NY