The History of the Table
 Mendeleev's Periodic table in 1869
Mendeleev's Periodic table in 1869
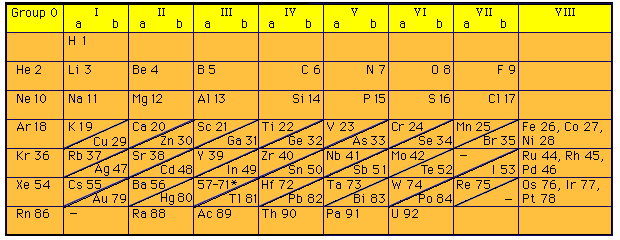
The first useable arrangement of the periodic table was in 1869, by Dmitri Mendeleev, a Russian chemist. He arranged the table by increasing atomic mass using the elements that have been found so far and left spaces for new discovered elements in the future. His table had mistakes in it, for example, the properties not being consistent between elements like Iodine and Tellurium. This became the layout for the future periodic table.
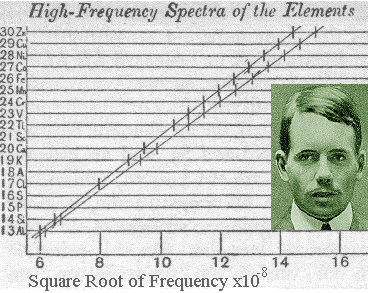
In 1913, Henry Moseley, an English physicist, published a paper that said that the frequencies in an x-ray spectra are equal to the atomic number of the elements. He believed that the properties of the elements would repeat periodically if they were arranged according to increasing atomic number. The modern periodic table is arranged by increasing atomic number as a result of Moseley's law. The modern periodic law, or Moseley's law says that the properties of the elements are periodic functions of their atomic numbers.
In 1913, Henry Moseley, an English physicist, published a paper that said that the frequencies in an x-ray spectra are equal to the atomic number of the elements. He believed that the properties of the elements would repeat periodically if they were arranged according to increasing atomic number. The modern periodic table is arranged by increasing atomic number as a result of Moseley's law. The modern periodic law, or Moseley's law says that the properties of the elements are periodic functions of their atomic numbers.