Metals
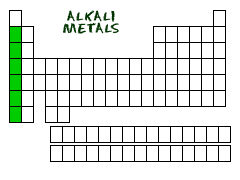
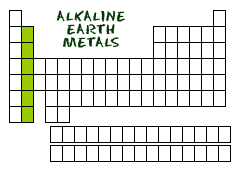
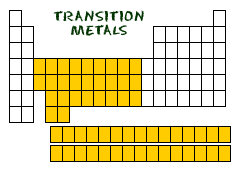
Metals make up most of the periodic table, being that 91 of the 118 elements are metals. They are located to the left of the stair-step-line that represents metalliods. Groups 1 and 2 of the table are the most active metals excluding Hydrogen. Going down a group, metallic properties increases. Metals are good conductors of heat and electricity, and are malleable, ductile, and have a metallic luster. They tend to lose electrons to create positive ions with a smaller atomic radius. Most metals are solid at room temperature, excluding mercury, a liquid.